CHANPREET S. answered • 04/17/22
Skilled and Experienced Tutor for Math and Chemistry
Hi Crystal,
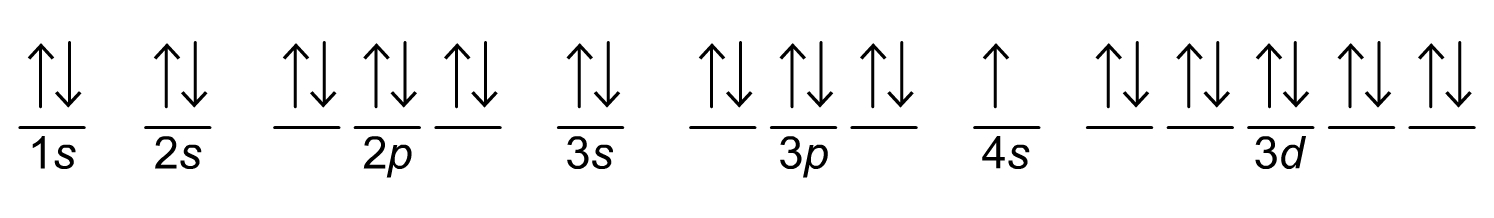
a) The full electron configuration for this representation here will be:
1s² 2s² 2p⁶ 3s² 3p⁶ 4s¹ 3d¹⁰
We write the orbital levels as they are arranged from left to right and then we count how many arrows we see (which indicate the number of electrons in each shell and orbital. This number of electrons is represented by the powers on the 1s, 2s, 2p etc.
b) To find what element is represented here, we can add all the exponent powers together. Adding them together gives us:
2 + 2 + 6 + 2 + 6 + 1 + 10 = 29
This will be the atomic number of the element. 29, from the periodic table, corresponds to Copper (Cu).
Hope this helps! Have a good one!