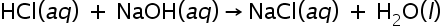Stanton D. answered • 02/04/20
Tutor to Pique Your Sciences Interest
Hi Commenwealth A.,
All of these chemicals dissociate in water except for water itself (that, only a little).
The (OH-) and the (H+) combine to make H2O. All other ions are spectators.
Since (Na+) and (Cl-) have almost no tendency to pull water apart and put things like (H+) or (OH-) onto themselves, there is no excess of (H+) or (OH-) present in the solution at the end of the neutralization reaction. That's the definition of "neutral" for aqueous solutions!
You might wonder why we get so excited about those levels of (H+) and (OH-) in water. It's because those particular ions have greatly destructive biological effects; they will tear your tissue right apart (on a microscopic scale). And they do comparably powerful effects on other chemicals.
Additionally, they represent the most powerful chemical species of their type (acid, base) which can exist in aqueous solution. Although there exist much more powerful acids and bases, that doesn't matter once they get into water, they simply pull water apart, and then only (H+) or (OH-) is the resultant species of note.
-- Cheers, -- Mr. d.


 ) reacts with a strong base (
) reacts with a strong base ( ) as shown below.
) as shown below.