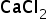Hatice B. answered • 02/03/20
PhD in Engineering with years of teaching and tutoring experience
molality is moles of solute divided by kg of solvent.
atomic mass of Ca: 40.078 g/mol
atomic mass of Cl: 35.453 g/mol
molecular mass of CaCl2 = 40.078 + 2*35.453
=110.98 g/mol
4.84 kg CaCl2 *1 mol CaCl2 / 110.98 g CaCl2 * 1000 g/1kg = 43.61 mol CaCl2
molality of CaCl2 solution = 43.61/13.5 = 3.23 mol/kg